Why review types matter
Evidence synthesis means combining insights from multiple studies to answer a research question. It’s how researchers build reliable knowledge, not from one study but from many.
Choosing the right review type is essential because each has its own structure, purpose, and level of rigour. A systematic review, for instance, is designed to answer a tightly focused question with strict methodology and is often used in medical research. A scoping review, by contrast, maps out what research already exists in a field, which is ideal when you’re still defining your question. (Covidence Blog).
| Type of Review |
Description |
| Systematic Review |
Systematic reviews are the best known type of review in certain fields. They aim for exhaustive, comprehensive searching and recommendations for practice.
Characteristics include:
- Draw together and appraise published knowledge within a particular area or topic.
- Adhere to set guidelines for conduct.
- Requires peer-review before publication.
|
| Scoping Review |
Scoping reviews provide an ‘environmental scan’ (preliminary assessment) of the literature that is currently available in an area of research.
Scoping reviews differ from mapping reviews as the outcome is only the review, not to conduct further reviews or research.
Characteristics include:
- Be a quick search than can inform if a full review is needed.
- Systematic approach.
|
| Literature Review |
Characteristics include:
- Establish a theoretical framework for your topic / subject area.
- Define key terms, definitions and terminology.
- Identify studies, models, case studies etc supporting your topic.
- Define / establish your area of study, i.e. your research topic.
|
| Integrative Review |
Characteristics include:
- Generate or refine a theory or hypothesis.
- Combine empirical and theoretical research.
- Examine research on a given health phenomenon.
- Inform healthcare policy and practice.
|
| Meta-Analysis |
Meta-analysis provides statistical combination of the results of quantitative studies. They aim for exhaustive, comprehensive searching. Meta-analysis can be included in systematic reviews but note that not all systematic reviews contain a meta-analysis.
Characteristics include:
- Able to draw together smaller studies to contribute to larger picture of impact.
- Time-efficient for decision makers, compared with reviewing individual studies.
|
| Qualitative Review |
Qualitative reviews are interpretative studies that can incorporate reports from users and observations from practitioners to allow for broader understanding than data-only would allow.
Characteristics include:
- Allows for synthesis of non-statistical information.
- Can highlight themes across individual studies.
- Complements research evidence with reports from users and observations from practitioners.
|
Adapted from University of Newcastle, Systematic review guide
Systematic review
Systematic reviews identify areas of uncertainty and where little or no relevant research has been done and where new studies might be needed.
A systematic review is:
- An authoritative account of the existing evidence using reliable, objective, thorough and reproducible research practices.
- A method of making sense of large bodies of information and assists in determining what works and what doesn’t in healthcare.
It’s a good idea to familiarise yourself with the systematic review process before beginning your review. You can do this by searching for other systematic reviews to look at as examples and by learning how to distinguish between types of reviews.
What are systematic reviews? (Cochrane video)
Other review types
Sutton, A., Clowes, M., Preston, L. and Booth, A. (2019), Meeting the review family: exploring review types and associated information retrieval requirements. Health Info Libr J, 36: 202-222. https://doi.org/10.1111/hir.12276
Systematic review types: meet the family (Covidence)
Literature review
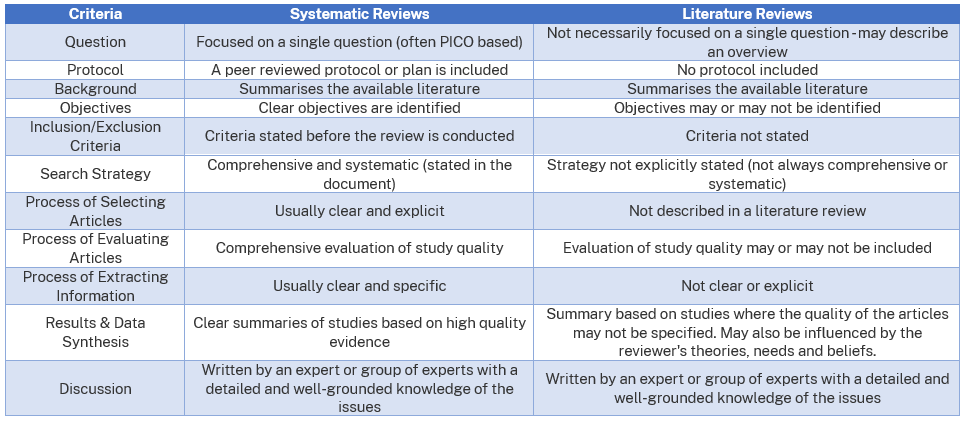
A literature review summarises, interprets, and critically evaluates the literature to establish current knowledge of a subject.
Both review types synthesise evidence and provide a summary of information. The key difference lies in the methodology. Systematic review methods are transparent and reproducible, designed to minimise bias. Unlike systematic reviews, the methods are generally not predefined and are not described in detail in the review. The search may be comprehensive but is not designed to be exhaustive.
How does a Systematic Review differ from a literature review?
 Action items
Action items